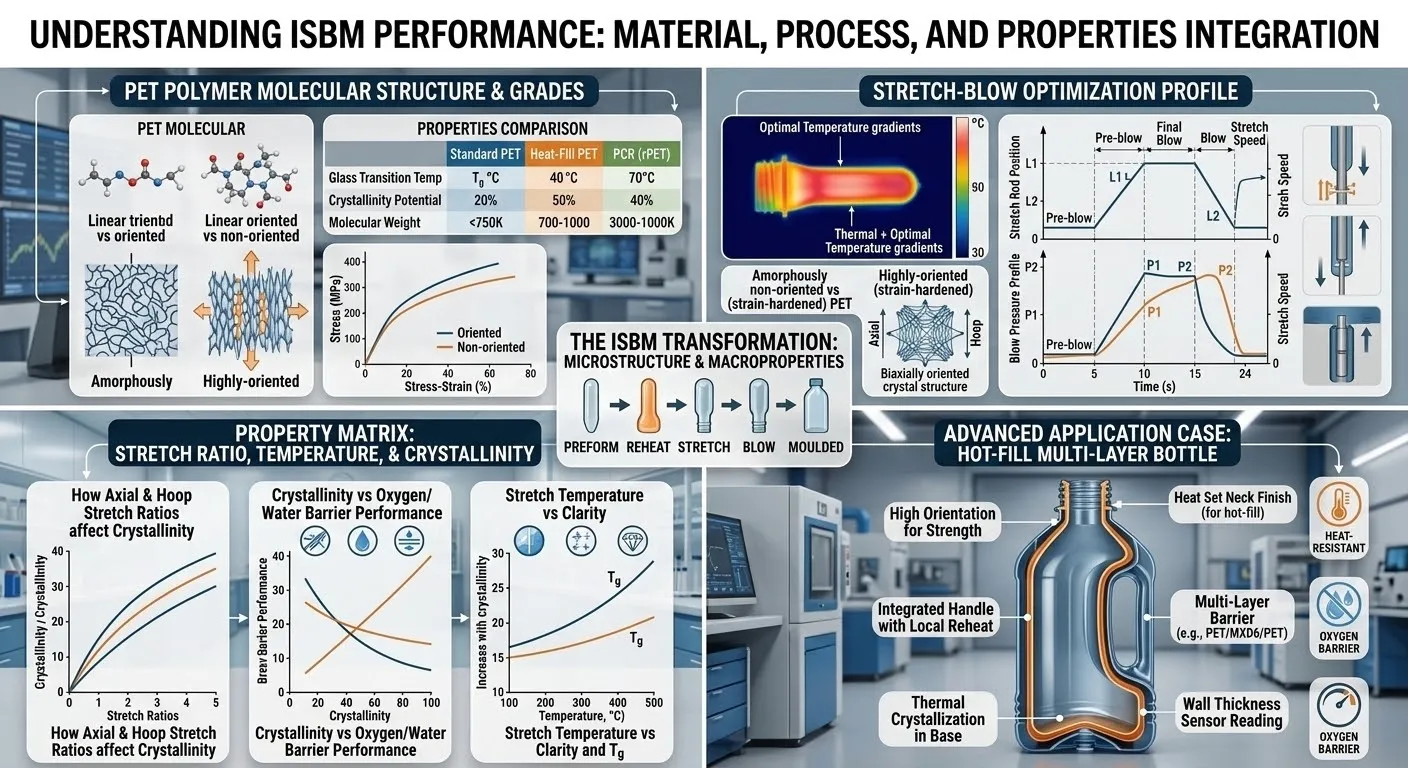
Are ISBM machines suitable for cleanroom production? A Deep Engineering Guide to Sterile Medical Packaging
Within the highly sophisticated and relentlessly regulated ecosystem of global plastic packaging manufacturing, absolutely no sector commands a higher degree of engineering scrutiny and environmental control than the production of medical devices, sterile pharmaceuticals, and premium biological cosmetic containers. In these life-critical industries, a plastic container is far more than a mere physical vessel; it is the ultimate biological barrier, the guarantor of chemical stability, and the absolute protector of product purity. Consequently, these high precision containers must be manufactured inside profoundly controlled, ultra-sterile environments known as cleanrooms. When multinational pharmaceutical giants, elite life science corporations, and global aseptic filling facilities engage in deep technical consultations with エバーパワー—an authoritative Brazilian Injection Stretch Blow Molding equipment manufacturer dominating the South American and international supply chains—the most critical and non-negotiable assessment is always: Are ISBM machines suitable for cleanroom production?
From an advanced engineering perspective, the definitive answer is an absolute yes, but this confirmation comes with a severe technical caveat: success requires the deployment of heavily customized, architecturally reimagined, top-tier ISBM platforms. Traditional heavy industrial plastic machinery, characterized by inherent hydraulic oil emissions, high-frequency mechanical friction, and uncontrolled pneumatic exhaust turbulence, acts as a natural destroyer of cleanroom environments. However, through the relentless integration of fully electric servo-driven technology, oil-free self-lubricating mold engineering, and highly intelligent pneumatic exhaust management systems, modern single-stage ISBM machines have flawlessly evolved into the ultimate medical manufacturing engines, fully compliant with ISO Class 7 and even higher aseptic standards. In this exhaustively detailed, highly authoritative technical whitepaper, we will completely deconstruct the microscopic physics of cleanroom ISBM manufacturing. We will deeply analyze the eradication of mechanical particulate contamination, explore the absolute supremacy of full-servo architectures, and demonstrate precisely how the world-class Ever-Power machinery matrix empowers your facility to dominate the high-value medical packaging market with zero-defect, zero-contamination perfection.
Phase One: The Physical Challenges of Cleanroom Standards versus Hydraulic Flaws
To truly comprehend why only a highly select echelon of ISBM machines is granted clearance to enter a sterile manufacturing suite, one must first profoundly understand the unforgiving physical requirements of a cleanroom environment. The International Organization for Standardization imposes brutally strict limitations on air purity. For instance, within an ISO Class 7 cleanroom, the total volume of suspended microscopic particles larger than half a micron per cubic meter of air is strictly capped. Even the most minuscule speck of mechanical dust, or a microscopic, invisible aerosol droplet, will instantaneously trigger cleanroom environmental monitoring sensors, leading to the immediate quarantine and catastrophic financial write-off of an entire batch of immensely expensive pharmaceutical packaging.

Under these draconian atmospheric standards, traditional fully hydraulic injection stretch blow molding machines expose their fatal manufacturing flaws. Hydraulic systems rely upon hundreds of liters of high-temperature hydraulic fluid to transmit the massive kinetic energy required for mold clamping and polymer injection. During the relentless, high-pressure, high-frequency operation of the machine, it is a physical inevitability that microscopic amounts of hydraulic fluid will seep from cylinder seals, manifold blocks, and complex hose fittings. More devastatingly, as this scalding fluid circulates throughout the system, it vaporizes to form micrometer-sized oil aerosols. The moment these oil mists are released into the cleanroom atmosphere, they not only adhere to and permanently destroy the expensive High-Efficiency Particulate Air (HEPA) filtration arrays, but they directly contaminate the interior walls of the transparent polymer containers being formed. Such hydrocarbon contamination guarantees immediate failure during United States Food and Drug Administration or European Medicines Agency biological compatibility audits.
Phase Two: The Core Engine of the Sterile Revolution Full Servo Architecture
To completely eradicate the catastrophic contamination risks posed by traditional hydraulic oil mists while simultaneously achieving sub-millimeter precision over mechanical kinematics, Ever-Power spearheaded an industry-shattering thermodynamic revolution: the absolute transition to fully electric servo-driven architectures. Servo technology is not merely a tool for accelerating production velocity; it is the sole, universal master key to unlocking medical-grade sterile manufacturing capabilities.
Within our elite cleanroom-ready platforms, such as the globally celebrated EP-HGY150-V4-EV フルサーボ4ステーション射出延伸ブロー成形機 そして超精密な EP-HGY50-V3-EV フルサーボ射出延伸ブロー成形機 designed specifically for extremely space-constrained environments producing premium ophthalmic dropper bottles and specialized vials, the massive and contamination-prone hydraulic reservoirs have been entirely eliminated. Every single critical motion axis of the machine—including the ultra-high-pressure injection carriage, the immensely heavy tie-bar clamping mechanism, the high-frequency rotary indexing table, and the microscopic stretch rod descent system—is directly commanded by top-tier, fully enclosed electromagnetic servo motors.
This fully electric architecture does not merely deliver “zero hydraulic oil” emissions to prevent aerosol contamination; it introduces an astonishing degree of electrical silence and operational stability. Servo motor actuation does not generate the violent concussive shocks and low-frequency vibrations characteristic of hydraulic directional valves, thereby drastically reducing the shedding of microscopic metallic dust caused by mechanical resonance. This absolute pinnacle of operational cleanliness solidifies the Ever-Power full-servo series as the ultimate platform of choice for manufacturing high-value blood plasma containers, high-precision in-vitro diagnostic reagent bottles, and premium aseptic culture media packaging.
Phase Three: Tooling Engineering for Contamination Prevention Self-Lubricating Metallurgy
Ensuring the cleanliness of the machine chassis completes only half of the sterile manufacturing equation. In the single-stage ISBM process, the custom tooling is the direct physical medium that interfaces with the scalding polymer melt. If the internal engineering of the mold violates aseptic protocols, the overarching cleanliness of the machine itself is rendered entirely useless. Traditional tooling incorporates a vast array of sliding mechanical components, including guide pillars, angled cam pins, ejector plates, and sliding core blocks. These components historically depend upon the manual, scheduled application of thick industrial lubricating grease to prevent catastrophic metal-on-metal galling and seizure. However, under the intense thermal loads of continuous cleanroom operation, these greases rapidly carbonize, vaporize, or physically drip directly onto the transparent containers being formed, resulting in irreversible black speck contamination.
Operating as a totally vertically integrated manufacturer possessing a world-class, independent tooling research and development center, Ever-Power subjects our bespoke カスタムワンステップ射出延伸ブロー金型 to the most draconian cleanroom engineering standards on the planet. We have entirely eradicated the requirement for external applied greases. Every single sliding bearing, guide bushing, and friction surface within our cleanroom molds is machined from specialized, high-tensile bronze alloys deeply impregnated with solid graphite matrices. This advanced metallurgy allows the components to maintain an incredibly low coefficient of friction across hundreds of thousands of high-pressure clamping cycles by naturally shedding microscopic, solid dry-lubricant particles, absolutely guaranteeing zero liquid volatile emissions.
Furthermore, to actively combat the threat of iron oxide particulate contamination arising from mold surface oxidation, all tooling destined for medical or food-grade cleanrooms is carved exclusively from the highest industrial grade, corrosion-resistant, high-chromium martensitic stainless steel. The internal surfaces of the injection cavities, the hot runner manifold plates, and the blow mold cavities are subjected to extreme micro-level titanium nitride coating or flawless mirror-finish polishing. This immense attention to metallurgical detail not only ensures permanent corrosion resistance in highly humid cleanroom environments but imparts a flawless, pristine optical purity to the final medical containers.
Phase Four: Pneumatic Exhaust Isolation and Aerodynamic Airflow Management
A fundamental core of the ISBM process is the high-pressure stretch blow molding phase. To force the soft, pliable plastic preform to expand and perfectly replicate the mold cavity, the machine must instantaneously inject ultra-high-pressure compressed air, frequently exceeding thirty-five or forty bar. The microsecond this blowing action concludes, this massive volume of high-pressure gas must be rapidly exhausted from the machine to allow the mold to open. If this explosive blast of pneumatic exhaust is permitted to vent directly into the cleanroom atmosphere, the resulting violent air turbulence will instantly weaponize any microscopic dust settled on the floors or walls, throwing it into the air and completely destroying the meticulously engineered laminar flow of the facility’s air purification system.
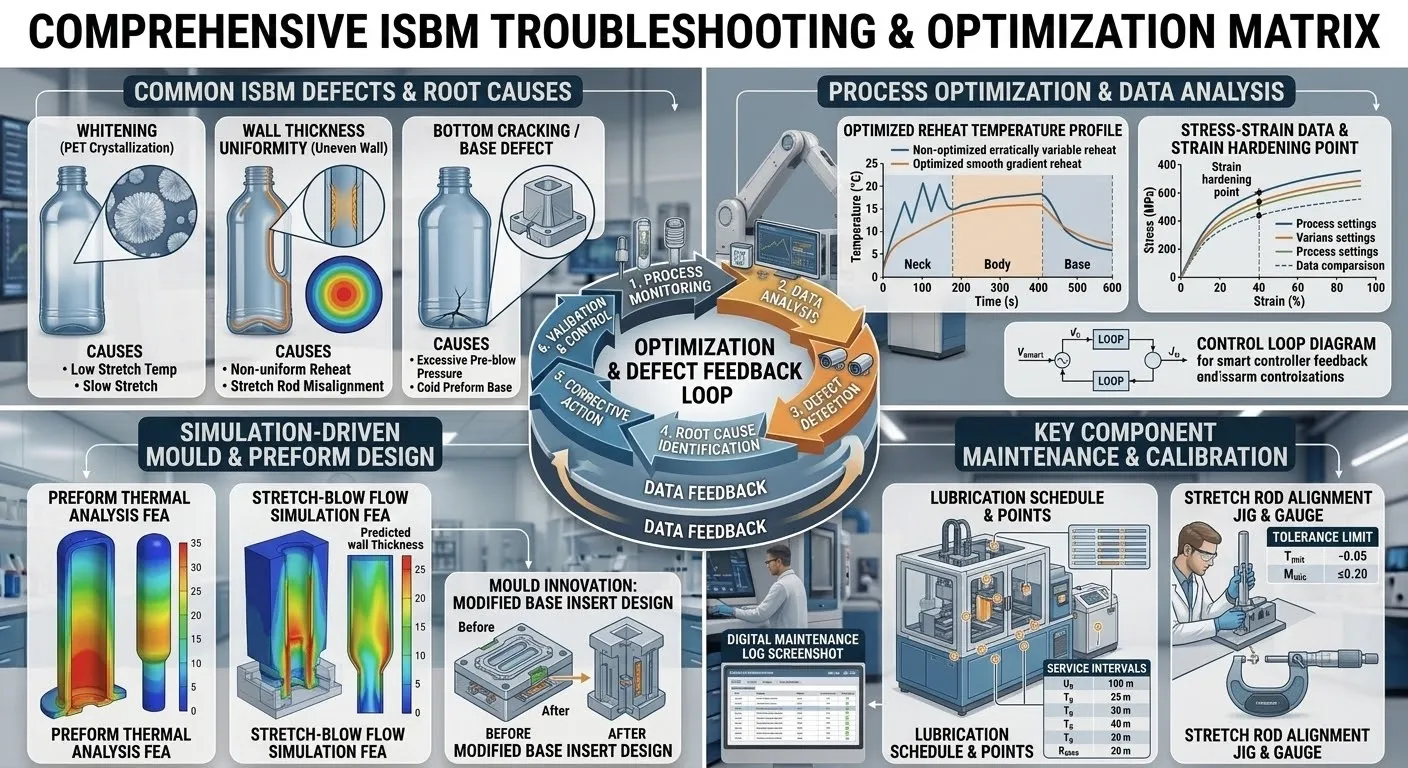
To seamlessly integrate our machinery into highly sensitive cleanroom aerodynamics, the engineering consortium at Ever-Power completely redesigned the pneumatic exhaust architecture. All high-pressure blow exhaust ports and low-pressure pneumatic cylinder exhaust valves are centrally channeled into heavily sealed, stainless steel collection manifolds. This waste gas is then directed through oversized, flexible sanitary ducting to be expelled entirely outside the cleanroom envelope or fed directly into the factory’s central exhaust recovery network. In addition to exhaust management, the actual compressed air utilized to blow inside the bottle is subjected to intensely rigorous, multi-stage Ultra-Low Penetration Air (ULPA) filtration and advanced cryogenic desiccant drying before it ever enters the machine’s primary intake, guaranteeing that the gas contacting the interior wall of the medical container is maintained at a state of absolute sterility.
Phase Five: Heavy Medical Production Scaling Capacity in Cleanrooms
When massive multinational pharmaceutical conglomerates demand the continuous production of hundreds of millions of large-volume Intravenous (IV) fluid bottles, sterile saline containers, or bulk parenteral nutrition packaging, the output capacity of standard single-row machinery is rapidly exhausted. However, physically deploying multiple production lines within a cleanroom requires astronomical capital expenditure due to the severe costs associated with expanding certified cleanroom floor space. To achieve an exponential explosion in production capacity without expanding the physical cleanroom footprint, Ever-Power’s Double-Row equipment architecture provides the ultimate economic solution.
Our leviathan-class flagship platforms, such as the incredibly powerful EP-HGY250-V4-B 2列4ステーション射出延伸ブロー成形機 そして EP-HGY200-V4-B 4ステーション射出延伸ブロー成形機, possess the engineering capability to inject and blow two parallel rows of medical-grade containers simultaneously within a single mechanical cycle. To specifically address rigorous cleanroom mandates, these double-row titans can be customized with high-strength physical isolation enclosures and overhead, roof-mounted Laminar Flow Hoods. This advanced aerodynamic design generates a continuous, downward-flowing curtain of highly filtered, positive-pressure sterile air directly over the core mold opening and closing zones. By artificially creating a localized, ultra-pure ISO Class 5 micro-environment within a broader ISO Class 7 cleanroom, we guarantee these large-volume medical containers receive absolute biological shielding during the highly vulnerable microseconds of mold ejection and transfer.

Phase Six: Agile Manufacturing Platforms for Diverse Medical Product Lines
For numerous modern life science companies and specialized medical device manufacturers, the primary challenge is not merely producing massive volumes, but managing an incredible diversity of product lines. They may be required to manufacture high-barrier, multi-layer ophthalmic dropper bottles on Monday, and seamlessly transition to producing ultra-clear laboratory centrifuge tubes by Thursday within the exact same cleanroom suite. In this highly flexible manufacturing environment, the ISBM machine must exhibit profound adaptability, rapid mold-changeover capabilities, and simplified sterilization maintenance protocols.
To conquer this agile medical manufacturing sector, Ever-Power provides a matrix of universally trusted, high-flexibility platforms. Models such as the highly acclaimed EP-BPET-125V4 4ステーション射出延伸ブロー成形機 そして EP-BPET-70V4 4ステーション射出延伸ブロー成形機 feature extremely accessible mold installation envelopes and highly intuitive operator interfaces. This heavily reduces the physical time technicians must spend inside the cleanroom performing complex mechanical changeovers, thereby mitigating the risk of human-introduced particulate contamination. For operations focused on geometrically straightforward medical containers that do not necessitate complex secondary thermal conditioning, our EP-BPET-94V3 3ステーション射出延伸ブロー成形機 offers a stroke of engineering genius. By entirely removing the thermal conditioning station, this architecture not only shrinks the physical footprint of the machine but drastically reduces the total number of internal moving parts. This fundamentally decreases potential particulate generation sources at the engineering root, making it the absolute ideal choice for balancing extreme cost-efficiency with uncompromising cleanroom purity.
Phase Seven: Conquering Extreme Medical Geometries The Six-Station Revolution
The medical sector frequently requires the production of highly specialized, ergonomically bizarre container designs. Examples include surgical drainage bottles with severely off-center aspiration nozzles, extremely flattened dialysis fluid pouches, or highly asymmetrical geometries designed to interlock with specific hospital diagnostic machinery. Attempting to blow these radically unbalanced containers on a standard four-station machine invariably leads to severe stress whitening, catastrophic wall-thinning, and unacceptable rejection rates.

To grant packaging engineers absolute, unbridled design freedom within a sterile environment, Ever-Power developed an unparalleled industrial marvel: the EP-HGYS280-V6 6ステーション射出延伸ブロー成形機. This unprecedented six-station platform provides immense, multi-stage thermodynamic reshaping capabilities for the most difficult-to-mold medical preforms. By executing incredibly delicate, directional heating and cooling across two fully independent thermal conditioning stations, even the most aggressively asymmetrical medical devices achieve flawless wall thickness distribution and immaculate optical transparency, easily satisfying the rigorous physical strength validations demanded by top-tier global medical brands.
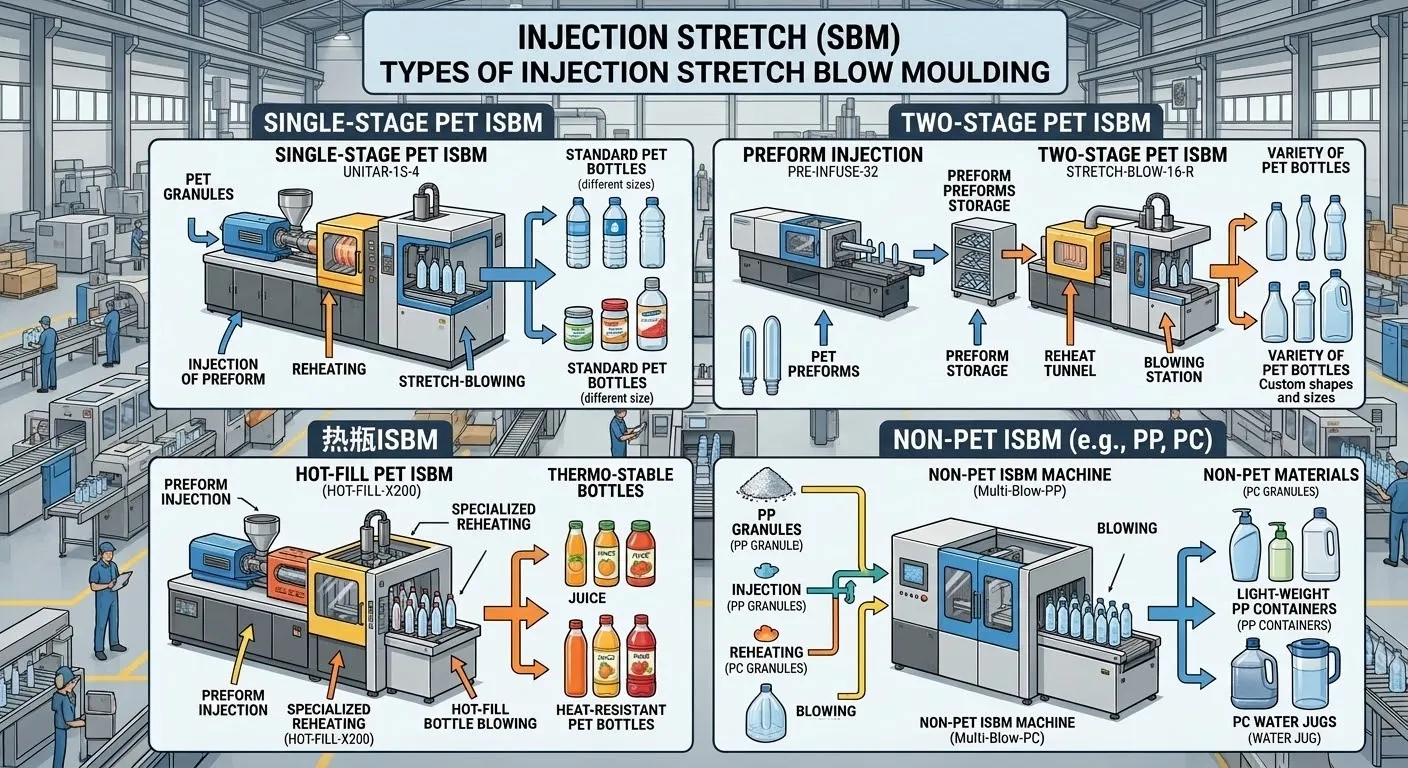
Phase Eight: Strategic Upgrades for Traditional Industrial Platforms
If your manufacturing facility has already heavily invested in our immensely powerful hydraulic-driven industrial workhorses—such as the heavily armored flagship EP-HGY650-V4 4ステーション射出延伸ブロー成形機, the high-performance EP-HGY250-V4 4ステーション射出延伸ブロー成形機, or the globally standardized EP-HGY200-V4 4ステーション射出延伸ブロー成形機 そして EP-HGY150-V4 4ステーション射出延伸ブロー成形機—and business expansion dictates a sudden pivot toward cleanroom production, Ever-Power provides profound, hybrid engineering retrofit strategies.
For these immense hydraulic platforms, we implement the strategy of Absolute Physical Isolation. Through advanced factory architectural planning, the main hydraulic power pack and primary pump systems—the sources of potential oil mist—are physically segregated into a heavily ventilated utility room located entirely outside the cleanroom boundary. Only the core structural chassis of the machine, containing the sterile mold platens and clamping mechanisms, is permitted to extend through a highly sealed, custom-engineered wall penetration into the cleanroom. Coupled with customized positive-pressure micro-environment enclosures over the mold area, even these monumental hydraulic beasts can be successfully tamed and perfectly integrated into high-level, aseptic medical manufacturing infrastructures.
Conclusion: Transcending Boundaries to Dominate Sterile Manufacturing
Thoroughly addressing the inquiry Are ISBM machines suitable for cleanroom production? is not simply a matter of verifying a technical specification; it is the ultimate litmus test determining whether an equipment manufacturer possesses the engineering fortitude to conquer the highest peak in the global polymer industry. In the unforgiving battlefield of medical devices and aseptic pharmaceutical packaging, any microscopic compromise—whether it be a single drifting particle of metal dust or an invisible wisp of hydraulic aerosol—will provoke devastating regulatory penalties and catastrophic brand damage.
As the undisputed industrial lighthouse headquartered in Brazil, projecting absolute engineering authority across the Americas and deeply serving the world’s most elite supply chains, Ever-Power categorically refuses to compromise on cleanroom standards. We deliver far more than machinery capable of extreme output velocities; we construct impenetrable aseptic fortresses that easily conquer the most draconian FDA and EMA regulatory audits. From the extreme purity of our fully electric servo drives to the microscopic precision of our self-lubricating, food-grade metallurgy and impenetrable pneumatic exhaust networks, the Ever-Power medical-grade machinery matrix will utterly obliterate your manufacturing risks, granting your corporation absolute, unyielding dominance in the world’s highest-value packaging markets.
Reconstruct Your Medical Manufacturing Empire Today
Do not allow highly contaminative, technologically obsolete hydraulic machinery to barricade your entry into the profoundly lucrative medical packaging sector. Forge a strategic alliance with the globally recognized cleanroom engineering leader, and deploy Ever-Power’s ultra-pure, zero-aerosol, full-servo intelligent manufacturing engines directly into your sterile suites to initiate a true era of zero-defect, uncompromising aseptic production.
Are you fully prepared to execute a completely comprehensive, highly rigorous industrial compliance audit of your cleanroom production strategy? Visit our global corporate headquarters at https://isbmmolding.com/, or immediately contact our elite medical engineering diagnostic team directly at [email protected] to secure a bespoke, cleanroom-certified equipment deployment blueprint and profound regulatory compliance consultation.